Have you ever wondered how they know what the atmosphere on Mars is made of? Did you ever doubt the nutritional information on the packaging of your favorite snack? If so, the tool that you would need to answer these questions is a mass spectrometer. Today’s blog provides a brief introduction to this tool, how they work and how ACER’s researchers are using it.
The mass spectrometer, or mass spec for short, has become an important tool in many aspects of science including genetics, biochemistry, pharmaceuticals, environmental science, geology and ecology. The mass spec is an instrument that tells us the masses of specific chemical elements in a sample. Briefly, a mass spec works by converting all of the chemical elements in a liquid, solid or gas sample to ions (‘ionizing’). The instrument then sorts or separates the ion based on their mass (specifically their mass to charge ratio) by applying a magnetic or electric field. A detector then records the specific ions present at specific times in the stream of ions.

While there are a number of different types of mass specs (accelerator mass spec (AMS), inductively-coupled plasma mass spec (ICP-MS), isotope ratio mass spec (IRMS) to name a few), but all mass specs include an ion source, a mass analyzer (which applies the magnetic or electric field), and an ion detector.
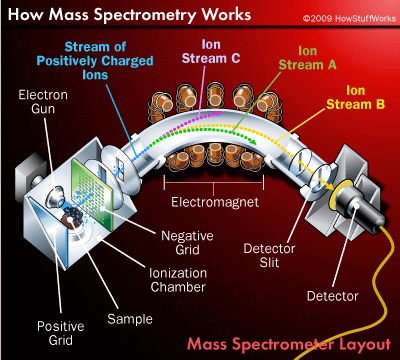
If you don’t remember your high school chemistry (or if that recollection is too painful to recall anything like it is for me), an ion is a charged (positive or negative) molecule. A mass spec creates these charged particles by firing electrons at the sample until it all breaks apart. The ions are then shot into an electric or magnetic field. This field causes the ions of different charge to move to the detector at different rates from the chamber where the field is applied. Just as a lighter box is easier to shove than a heavier one, lighter ions are deflected more than heavier ones and reach the detector first.
Returning to our examples, food scientists use mass spectrometry to determine nutritional information on the packaging of your favorite salty of surgery snack. A bag of potato chips, for example, a mass spec can tell me exactly how much sodium I will ingest per handful. And for the Martian atmosphere, the mass spec on the rover, Curiosity, has told us that the atmosphere is primarily CO2 and that we would not be able to survive without a supplemental supply of oxygen.
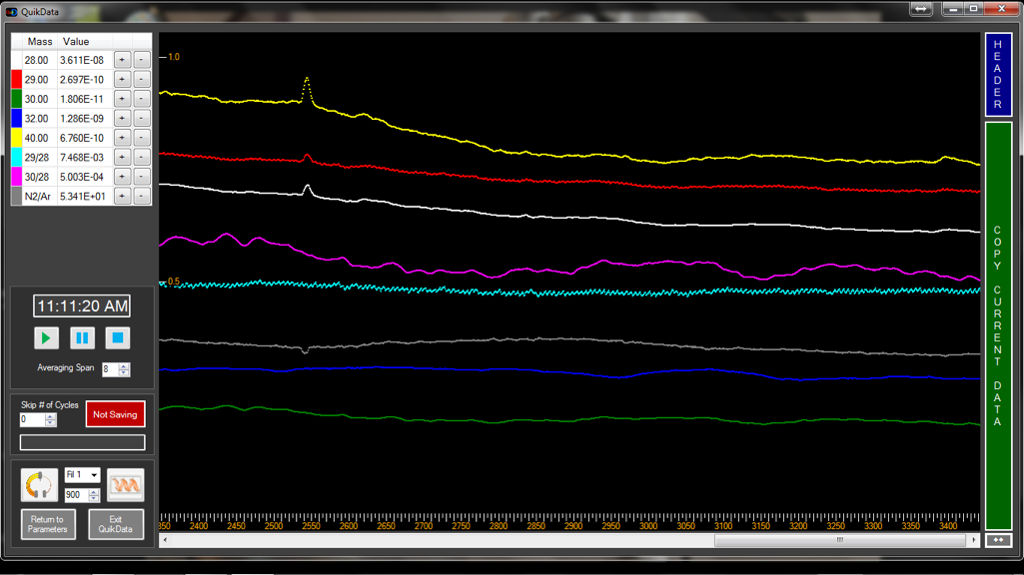
The photo above is a typical mass spec output. The ruler-like portion you see at the bottom simply gives the time (in seconds) at which the ion was detected. In the upper left hand corner you see a table listing mass and values. Colored lines on the plot correspond to a specific masses listed in this table. Looking closely at the colored lines, you see peaks along the relatively level lines. A peak indicates and ion and the height of the peak corresponds to its relative abundance. By comparing samples of known chemical composition along with these experimental samples, the relative abundance of specific elements can be determined.
ACER scientist, Dr. Behzad Mortazavi uses mass spectrometers in his work on nitrogen cycling in coastal ecosystems and is examining the degree to which oil impacted the ‘normal’ patterns of nitrogen cycling. One of the key processes in the nitrogen cycle is denitrification. Denitrification, the conversion of NO3 (and some related compounds) to nitrogen gas (N2) is carried out by specific groups of bacteria. In these times of increasing human impacts on coastal ecosystems, denitrification plays a critical role in ameliorating increased nutrient impacts on the coastal zone. Dr. Mortazavi’s results will indicate whether denitrification rates can be significantly changed by crude oil inputs and give us insight on some of the less visible impacts of the Deepwater Horizon oil spill. Mass specs are also being used in other ACER research. Master’s student, Emily Suebert, is using a mass spec to trace food web links to the top predators (e.g. sharks) along the northern Gulf of Mexico.